Medivew, Transformational Augmented Reality Medtech Company, Secures $9.9 Million Safe Funding
![]()
![]()
Investment will support key technology development, commercialization and regulatory submissions
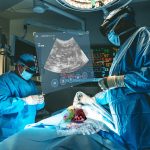
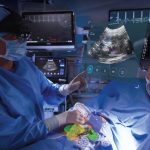
CLEVELAND, March 9, 2022 /PRNewswire/ — MediView XR, Inc., a Cleveland-based company, announced the closing of a $9.9MM round of SAFE financing syndicated by Inside View Investments, LLC, a private investment firm. MediView, a cutting edge medtech company, is combining augmented reality with surgical navigation to provide surgeons and healthcare professionals revolutionary 3D “X-Ray vision” through the skin for minimally invasive procedures.
“This round of financing provides a critical runway for MediView to achieve several milestones over the coming 12 months, particularly in the areas of FDA submissions, commercialization, strategic engagements, and investing in our expanding team and resources,” said Adam E. Rakestraw, MediView Cofounder & Chairman. MediView has raised approximately $14.4MM in total funding to date.
The company currently has three tiers of its platform in various stages of development.
The XR30, now in its early stages of commercialization, helps alleviate long-standing ergonomic challenges with medical imaging. Practitioners can use augmented reality to optimally position ultrasound imaging in the line of sight of their hands, tools, and the patient during minimally-invasive procedures. Additionally, XR30 enables collaboration between on-site and remote caregivers.
The XR50, to be submitted for FDA clearance in the first half of the year, adds procedural guidance capabilities to the XR30 by creating a visible pathway for the practitioner’s instrument during procedures.
The XR90 Augmented Reality Surgical Navigation System, MediView’s flagship product, is slated for FDA submission in the second half of 2022. XR90 empowers practitioners with “X-Ray vision” of the patient’s detailed internal soft tissue, organ, vascular and skeletal structures. The system further combines live imaging and tracking of the surgeon’s tools with the X-ray vision for comprehensive surgical navigation capabilities. Expanding access to less invasive cancer ablation therapies is the first application of the XR90 platform […read more ]